 |
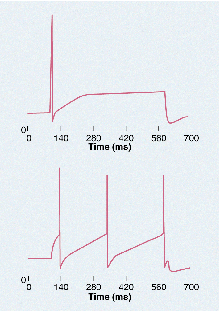
Excitable. Sympathetic neurons missing the M-channel (lower panel)
fire more than those that have the channel (upper panel).
CREDIT: DAVID MCKINNON |
Like people, neurons sometimes need to be steadied a bit so that they don't overreact to stimuli. That role is one of several that fall to potassium channels, tiny protein pores that allow potassium ions to flow out of neurons. So far, researchers have identified the proteins that make up most of the 20 or so known types of potassium channels. But one channel with a major influence on neuronal excitability, the M-channel, has remained mysterious--until now.
On page 1890, David McKinnon and Jane Dixon of the State University of New York, Stony Brook, and their colleagues report that they have identified the two proteins that together make up the M-channel. Their success is being heralded partly because it will help researchers understand how neural excitation is controlled. "This channel represents the most important regulator of excitability in many neurons," says University of California, Berkeley, neuroscientist Ehud Isacoff.
The M-channel may also be a key target for drug development. Even before the Stony Brook work, others had discovered that defects in the genes encoding the proteins cause a form of epilepsy. And M-channels are found in many brain areas including the hippocampus, where neural responsiveness can affect learning and memory. Knowing the identity of the channel's components will help researchers learn what turns it on and off and could lead to new drugs for epilepsy or Alzheimer's disease.
McKinnon and Dixon study sympathetic neurons, which control things
like heart rate and blood pressure. Like all neurons, sympathetic neurons
fire in response to signals arriving from other neurons, which open channels
that let positively charged ions flow into the cell. But some sympathetic
neurons are more excitable than others, firing many more action potentials
in response to a given stimulus.
 |
Excitable. Sympathetic neurons missing the M-channel (lower panel)
fire more than those that have the channel (upper panel).
CREDIT: DAVID MCKINNON |
In earlier studies, McKinnon and Dixon's team specifically tested neurons for the M-current, the flow of potassium ions across the membrane under conditions in which M-channels would be the only potassium channels open. They found that the less active neurons have M-channels while the more active neurons lack them. That made sense, because M-channels let positively charged potassium ions flow out of the neuron during the period leading up to an action potential. That reduces the neuron's excitability by countering the inward flow of ions triggered by neural signals.
The Stony Brook team used their knowledge of which neurons lack M-channels to help them search for the channel's protein components. In both types of neurons they screened through the RNA messages that indicate which proteins the neuron is making, to see whether any of the known potassium channel proteins were made only in the M-channel-containing neurons. KCNQ2, a potassium channel subunit that had not been linked to any known channel, fit the bill. In further experiments the researchers injected RNA encoding the different KCNQ subunits into frog egg cells and showed that KCNQ2 combines with another subunit, KCNQ3, to make a channel that behaves exactly like the M-channel.
This was not the first time the two proteins had attracted attention. Earlier this year, while the Stony Brook team was doing those experiments, a team at the University of Hamburg in Germany and another at the University of Utah reported that mutations in the genes that encode KCNQ2 or KCNQ3 cause a hereditary form of epilepsy. Finding out that the two proteins encode the M-channel "really makes sense," says Thomas Jentsch, a member of the Hamburg team, because "the M-channel has been shown to control neuronal excitability," and epileptic seizures occur when neurons become uncontrollably excited.
The prospect of controlling seizures via the M-channel already has drug company scientists intrigued. They "were all over the poster," McKinnon says, when he presented his team's work at the annual meeting of the Society for Neuroscience in Los Angeles last month. DuPont neuroscientist Barry Brown, a co-author on the paper, says drug companies can now use the subunits to screen drugs. "If you could find a drug that actually opened or enhanced the activity of M-currents, it may be a good antiepileptic drug," he says.
In addition, several compounds developed by DuPont as memory enhancers for Alzheimer's patients had already turned out to block the M-current. "That implies that the M-current is also involved in cognition," says Neil Marrion, a neuroscientist at the University of Bristol School of Medical Science in the U.K. "If you look at [animal models of] Alzheimer's, cell firing is actually dampened in the hippocampus," he notes. The cognition-enhancing drugs may work at least in part, he suggests, by "jazzing up" the excitability of neurons in this important memory area.
The DuPont drugs, along with the subunits and their genes, also provide a new set of tools for neuroscientists who study neural excitability. For example, the neurotransmitter acetylcholine enhances neurons' response to its excitatory signals by activating receptors that turn off the M-channel. But after years of research, no one has identified the intracellular messenger, triggered by acetylcholine, that turns the channel off. Having the subunits in hand "will help people to investigate what the messenger might be," says Marrion, who has studied the M-channel for a decade.
For instance, they can look for certain hallmark amino acid sequences
in the channel proteins that provide clues to the kinds of regulatory molecules
that act on the channel, mutate those amino acids to see the effects of
losing that regulation, and even study the effects of altered forms of
the M-channel in transgenic animals. "This work opens up whole new avenues,"
Marrion says.